Imagine this scenario: your team has spent years on research, the clinical trial has successfully passed all phases, and the investment is in the millions. Everything is ready for the international launch. You submit the dossier to the European Medicines Agency (EMA) and, suddenly, the process comes to a halt.
The reason? A terminological inconsistency in the translation of the patient information leaflet or a formatting error in the QRD template.
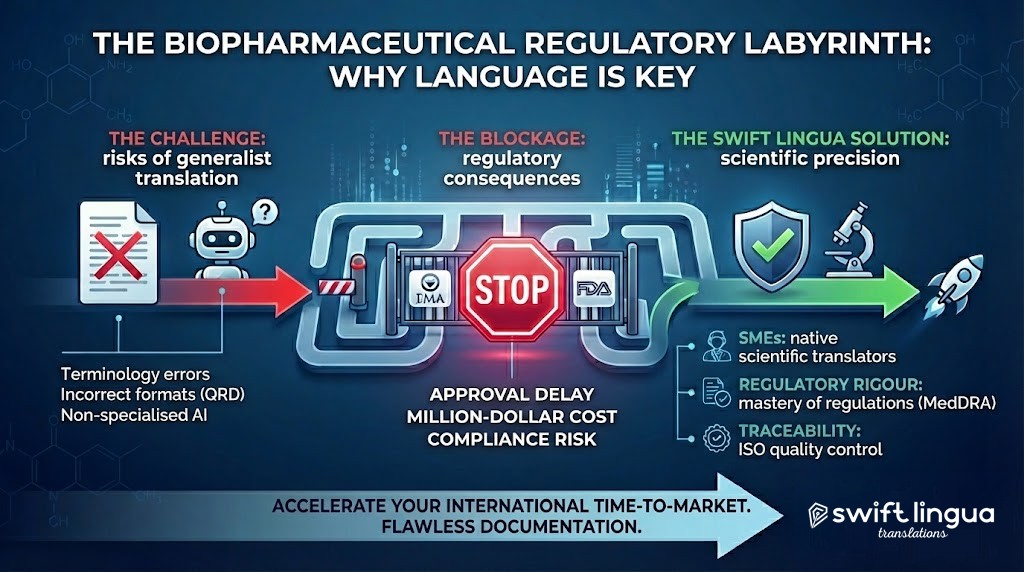
In the biopharmaceutical and Life Sciences sector, a translation error is not a simple “typo”. It is a regulatory block that delays your time-to-market by months, makes costs skyrocket, and puts regulatory compliance at risk.
Science does not allow for ambiguities (and neither do regulatory agencies)
Submitting the documentation for a new drug, veterinary product, or medical device means facing a true regulatory labyrinth. Bodies such as the EMA in Europe or the FDA in the United States demand millimetric precision.
At this level of demand, vocabulary and format are the law. Working with non-specialised machine translation tools or generalist agencies means taking on an unacceptable clinical and legal risk. Conventional artificial intelligence does not understand posology, is unaware of pharmacovigilance protocols, and does not know how to interpret strict local labelling requirements.
When it comes to health, there is no room for interpretation or literary creativity. Only absolute accuracy will do.
The Swift Lingua methodology: Translating science with scientific rigour
At Swift Lingua, we know that the true luxury for a Regulatory Affairs Manager is not just meeting deadlines, but being able to sleep soundly knowing the technical documentation is flawless.
That is why our approach for the pharmaceutical and biotechnology industry (with special attention to the powerful hub at our headquarters in León and nationwide) is based on an unbreakable safety net:
- SMEs (Subject Matter Experts): Your texts do not pass through the hands of generalist linguists. We work exclusively with native translators who hold higher education degrees in health sciences, medicine, biology, or pharmacy. They understand the science behind the words.
- Mastery of Regulatory Rigour: Our team is constantly updated on local and international regulations. We master the use of standardised dictionaries (such as MedDRA) and specific formatting requirements (QRD templates, instructions for use or IFUs).
- Traceability and Quality Control: We implement workflows with double and even triple review (ISO certification), guaranteeing that the term used on page 2 is exactly the same as on page 200.
Right-first-time compliance: Your Time-to-Market secured
The ultimate goal of our work is not to hand you a document in another language, but to pave the way for approval.
Avoiding endless rounds of questions and corrections from health authorities accelerates your international Time-to-Market. It allows you to focus your resources on what truly matters: continuing to innovate and improving patients’ lives.Is your biopharmaceutical company preparing its next international launch? Do not let language be a barrier. Contact the Swift Lingua team and let’s talk about how to bulletproof your clinical documentation.